Research
Cilia-Cilia Joining
A remarkable but poorly understood phenomenon occurs in certain C. elegans sensory neurons: adjacent cilia physically join tip-to-tip, forming elongated inter-ciliary bridges. This cilia-cilia joining process — also called juxtaposed cilia-cilia elongation — represents a novel mode of ciliary remodelling whose molecular basis and physiological significance have remained almost entirely unexplored. The Kaplan Lab is one of the few groups in the world dedicated to understanding this phenomenon.
What is cilia-cilia joining?
In the amphid sensory organ of C. elegans, cilia from neighbouring neurons come into contact and undergo a regulated elongation process in which the membranes of two adjacent cilia fuse or tightly associate. The resulting structure extends beyond the normal cilia length and requires an intact BBSome and specific regulatory GTPases. Disruption of joining correlates with sensory deficits, suggesting a functional role in inter-neuronal communication or sensory integration.
Key findings
Open questions
The lab is actively investigating: (1) which membrane fusion or tethering factors mediate the initial contact between cilia; (2) whether joining is regulated by neuronal activity or environmental cues; (3) whether analogous structures exist in vertebrate sensory organs; and (4) whether mutations in joining-related genes contribute to undiagnosed ciliopathy phenotypes in human patients.
Methods
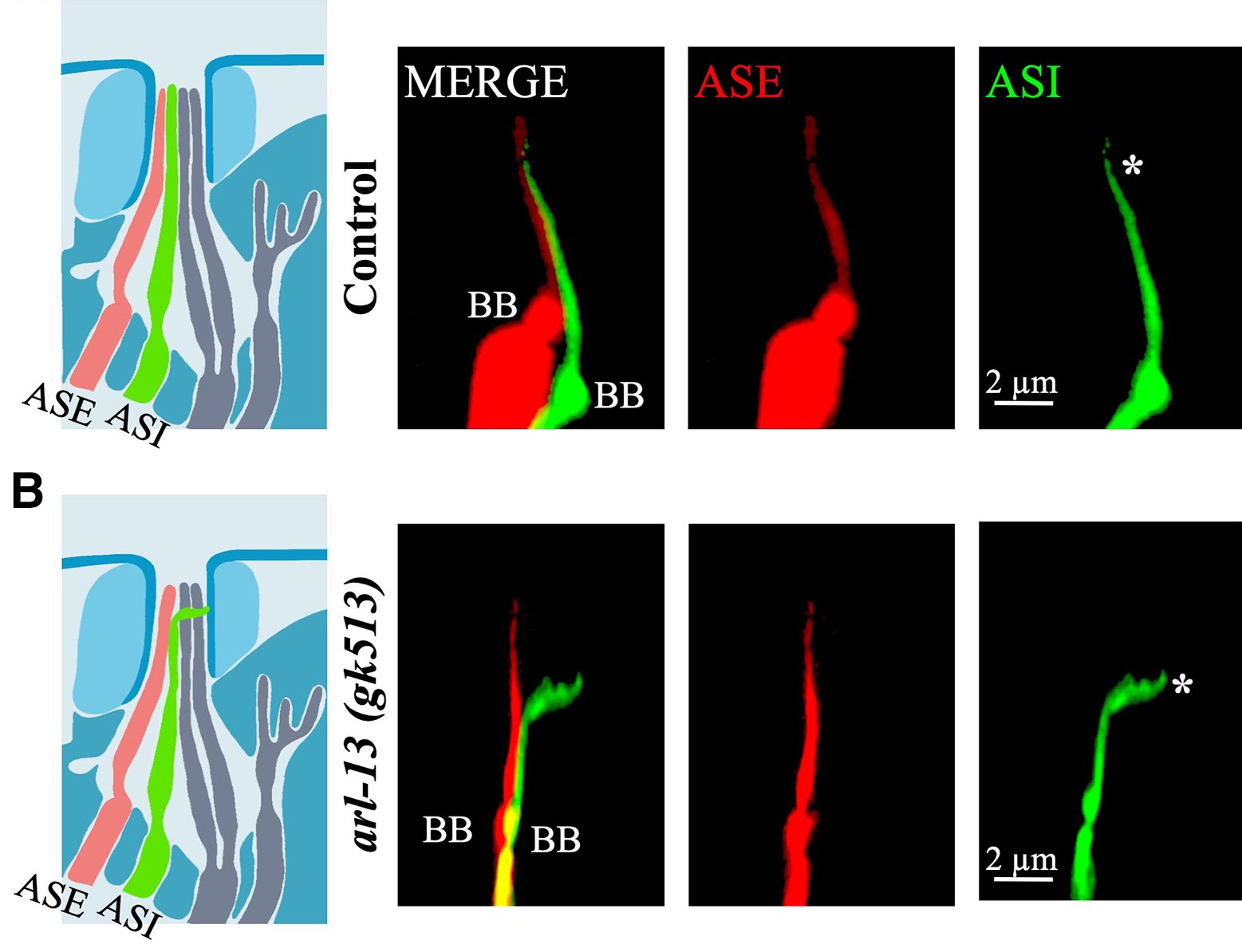
Juxtaposed cilia-cilia elongation in C. elegans sensory neurons.
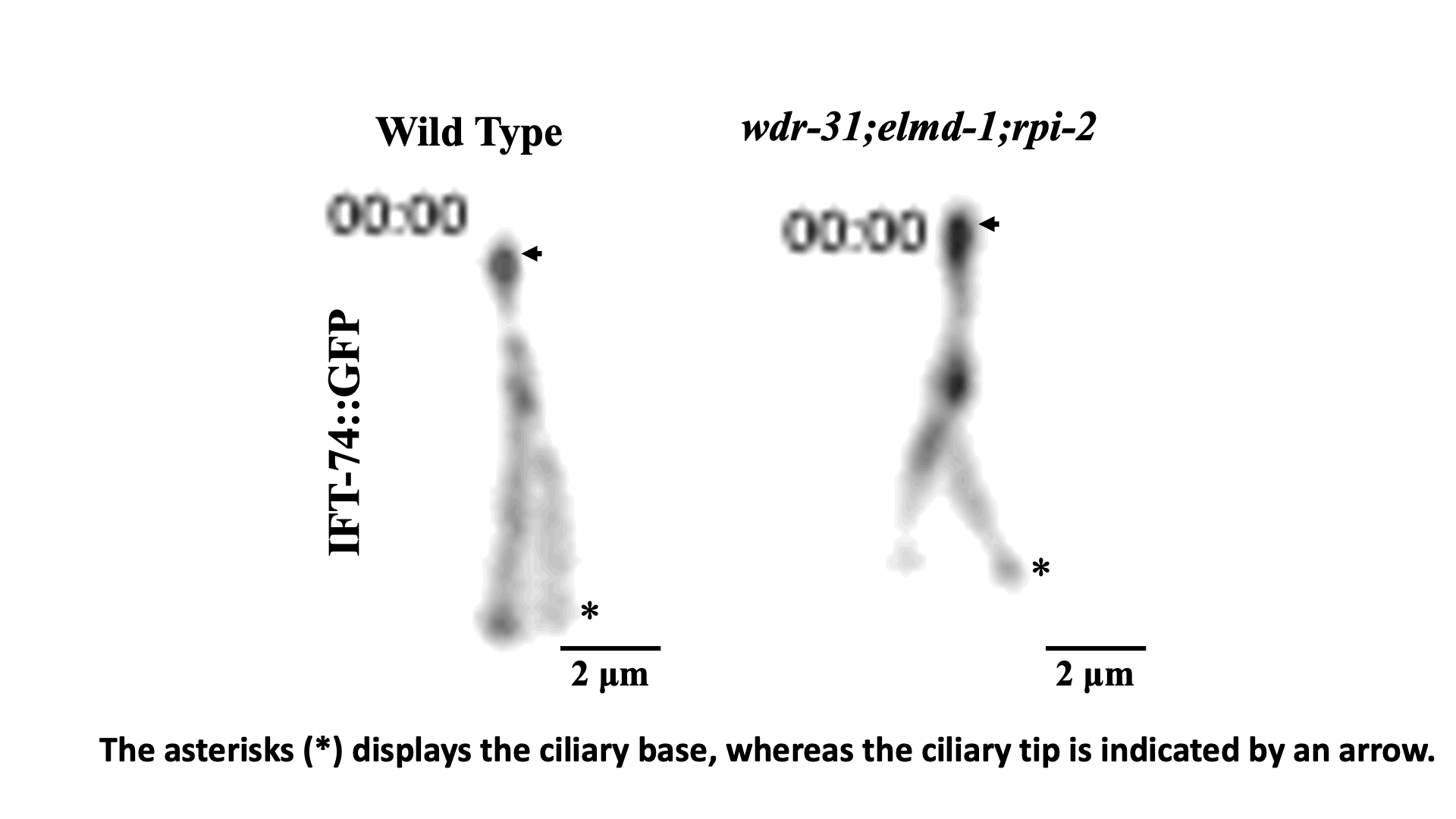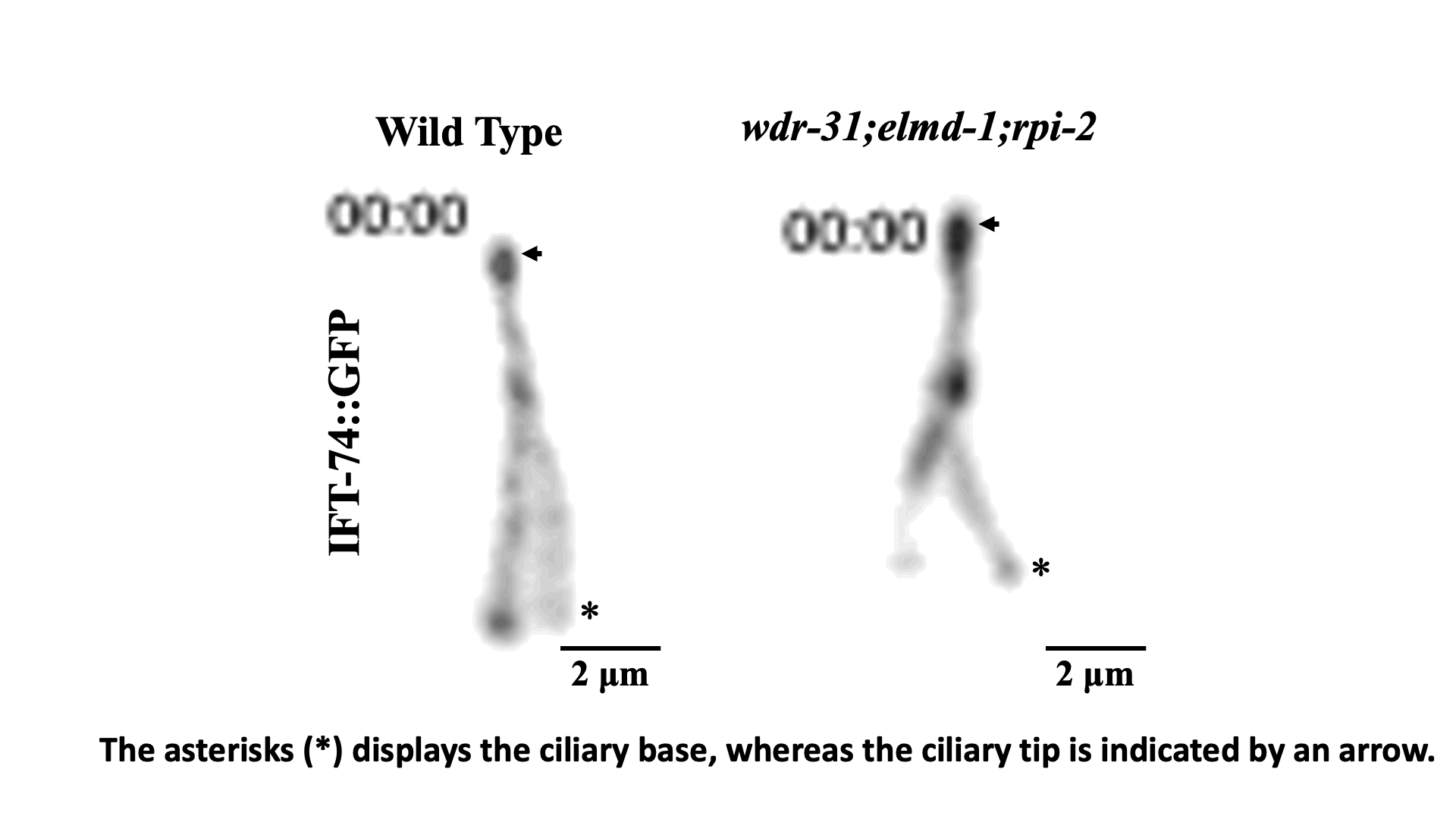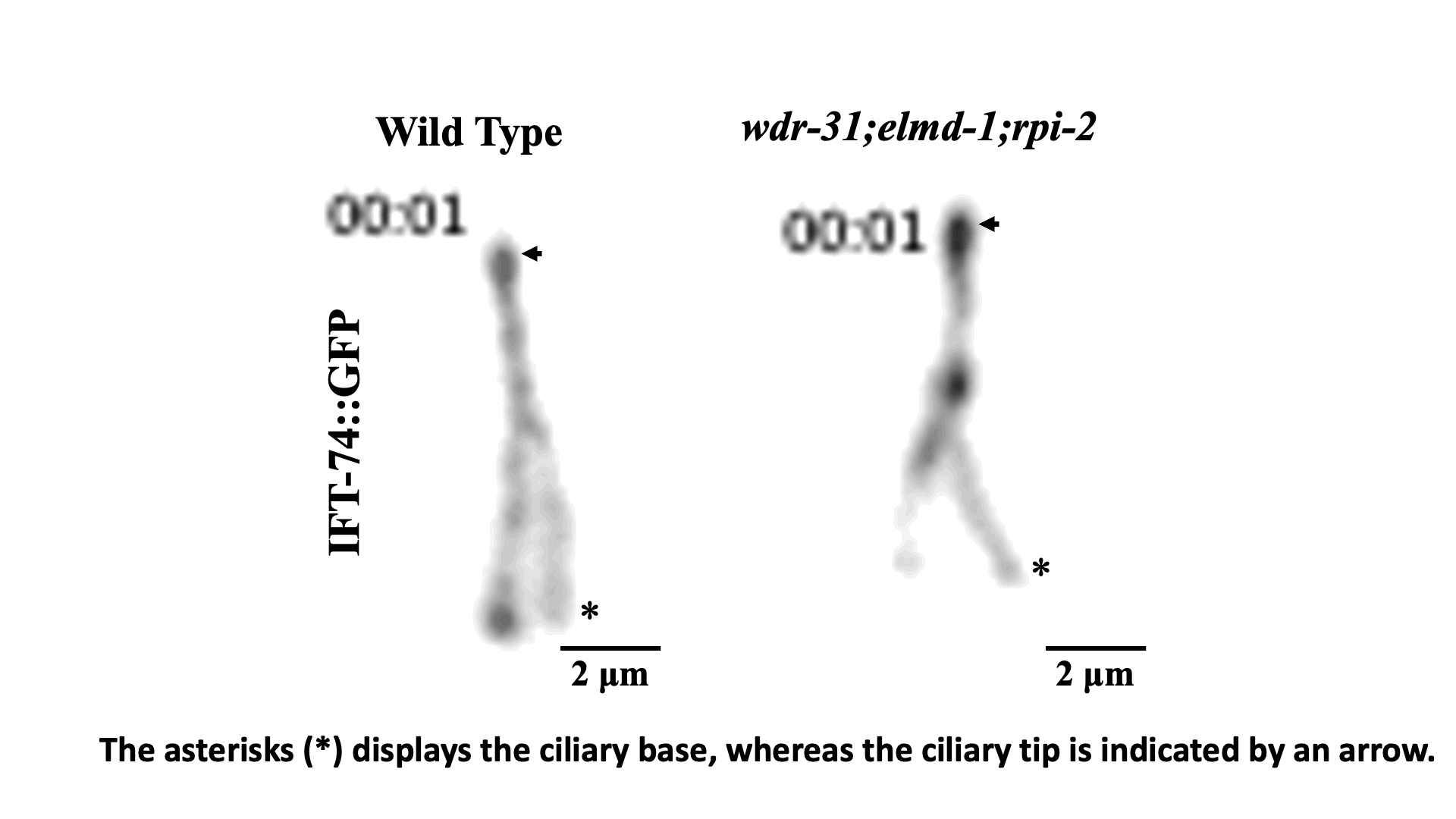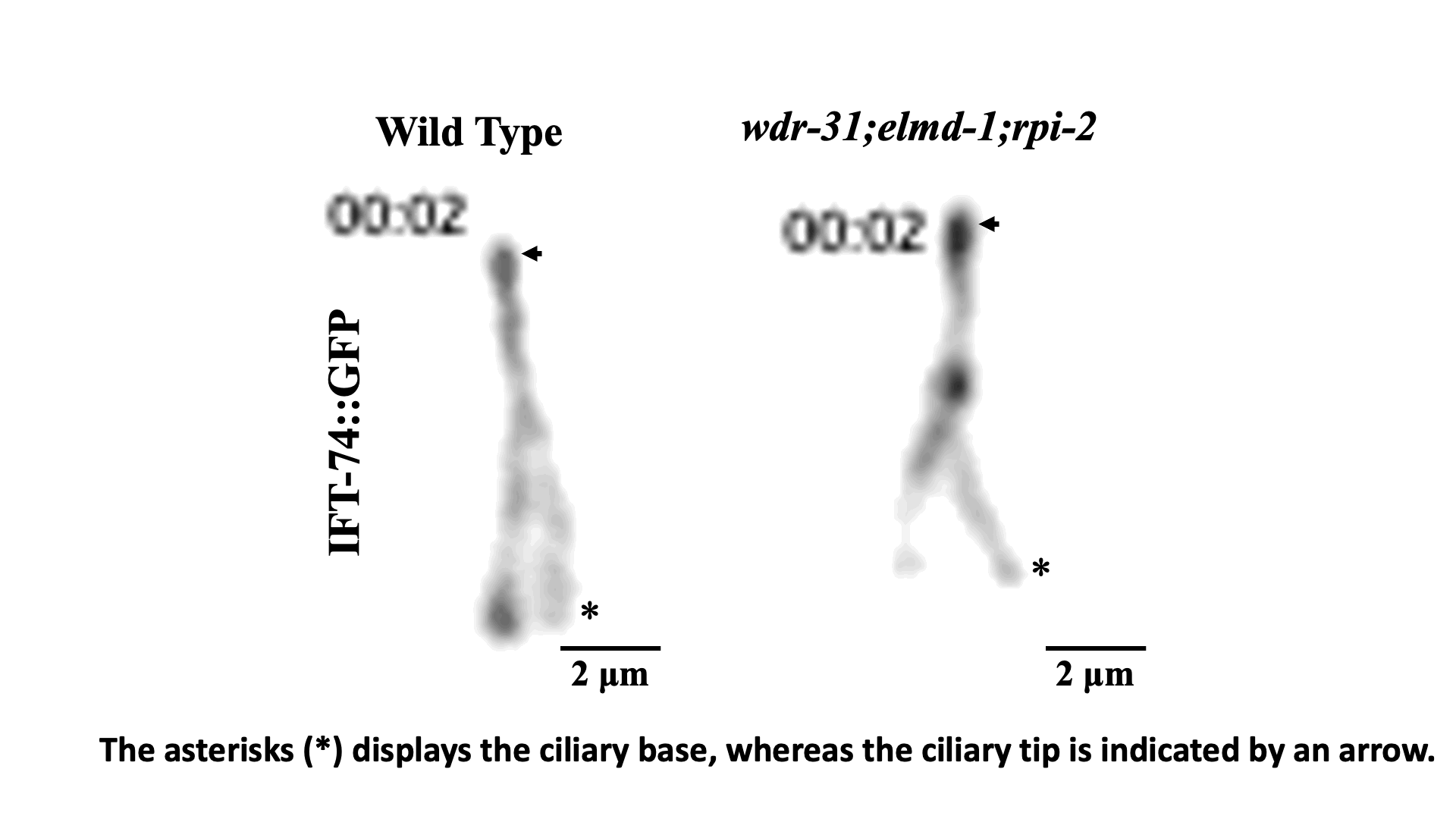
IFT train dynamics imaged by TIRF microscopy. Turan et al., STAR Protoc 2022.